
In his model, Neil Bohr compared the model of the atom with the model of planets and the sun. An atom may have a large amount of space as most of the particles passed with small deviations through it.Also, the positive charge has a very minute size in the atom. There is a positive charge (nucleus) in the atom which contains most of the mass of the atom.

However, few particles reflected over 90 degrees, depicting the presence of highly dense material at the centre. However, he found that most of the alpha particles passed. In the experiment, Rutherford thought that as per Thompson’s model all the alpha particles would pass through the foil. In his experiment, Ernest Rutherford used a gold foil on which he passed alpha beams which are beams from helium nuclei. Ernest Rutherford model (Gold Foil Model) After which, he postulated the plum pudding model of the atom in which electrons and protons (the positively charged matter) are unevenly placed in a pudding. To verify it he then used magnets instead of plates, in which he found the mass to charge ratio of the deflected particles which showed the particles that are deflected have very less mass than an atom. He found that the rays are getting deflected toward the positively charged plate or away from the negatively charged plate (depicting the deflection of electronics). And he placed two opposite charged electric plates for the rays to get deflected. In his experiment, Thompson used an anode and a cathode, where the cathode emitted the ray and the anode received it. JJ Thompson did a cathode ray tube (CRT) experiment in which he found the existence of electrons in the atom.Ī cathode-ray tube is a packed tube having a vacuum inside it with a sufficiently needed flow of cathode-ray inside it used by Thompson Similar to the discharge of lightning.

A chemical reaction is simply a combination or rearrangement of various atoms in different conditions.A compound is formulated using a combination of atoms of either the same or different atoms.An element has similar or identical atoms.Matter is made up of various tiny invisible particles commonly known as atoms.
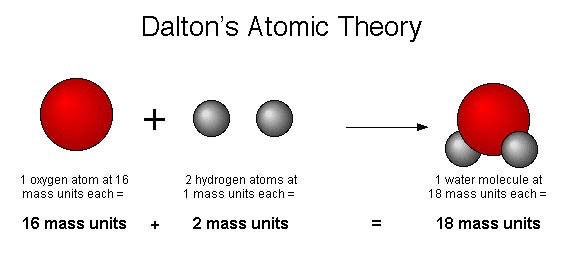
The main postulates of Dalton Atomic theory :. And also hypotheses about the difference in masses due to the difference in the atomic structure of the elements. And found that they show a common pattern in their formation or combinations. This was the first scientific hypothesis of the atom, considering Dalton arrived at his findings by testing and empirical investigation of the data.ĭalton came to his conclusion by studying various masses of various elements such as tin, oxygen and their combinations. Dalton claimed that every compound is made up of atoms of a unique, distinct kind, which can unite to build more complicated structures even though they can’t be transformed or damaged chemically or physically. It later became the fundamental building block of atomic structure or chemical structure. Dalton’s Atomic Theoryįrom 1743-to 1794 Dalton created his theory referring to Antoine Lavoisier’s theory using the conservation of mass and definite proportion reference. Here, in this article, we’ll look at Dalton’s atomic structure and how he found the atomic structure with some other scientists’ models such as the JJ Thompson atomic model, Rutherford atomic model, etc. And if we speak about the atom Democritus who was a Greek philosopher was the first person to use the term atom. Moreover, the commonly and a very famously known Dalton’s atomic theory is one of his contributions. However, an English scientist, meteorologist and physicist named John Dalton first mentioned that everything is made up of atoms in the book he published in 1808. Atoms have protons and neutrons in the center, making the nucleus, while the electrons orbit the nucleus.Many researchers and thinkers, notably Newton, Galileo, Lavoisier, and Boyle, explored and expanded on the notion of atomic structure and atoms.
